MICRONEEDLING WITH SKIN PEN FOR ACNE SCARS AND NECK WRINKLES
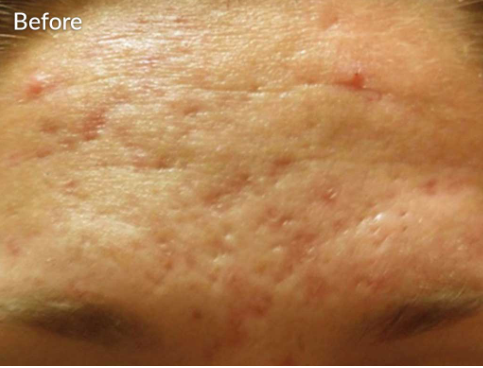

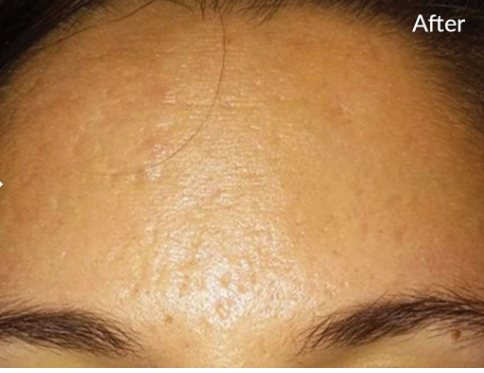
DO YOU HAVE ___________?
- Acne Scars?
- Wrinkles on your neck and below your chin?
- Skin depressions from previous acne breakouts?
- Discoloration due to previous acne breakouts?
- Lines on neck?
YOU MAY BE INTERESTED IN MICRONEEDLING WITH SKINPEN
SkinPen creates hundreds to thousands of “micro” skin punctures per second to stimulate the skin’s natural wound healing process – inflammation, proliferation, and remodeling – to prompt tissue remodeling without causing scar tissue formation. Most patients can return to normal activities within 24 hours.
HOW IS SKNPEN DIFFERENT FROM OTHER SKIN TREATMENTS?
Unlike some alternatives, SkinPen has been clinically proven to be effective for women and men with with all Fitzpatrick Skin Types I – VI. Likewise, unlike lasers or chemical peels that can damage skin over the long term, SkinPen treatments, when properly spaced and overseen by a physician, can be used for years.
WHY USE SKINPEN MICRONEEDLING?
IS SKINPEN MICRONEEDLING SAFE?
Yes, by design. SkinPen’s patented – and single-use – sterile needle cartridge is built with safety in mind. SkinPen is also surrounded by a custom-designed BioSheath that acts as a barrier to prevent cross contamination between procedures. That’s part of the reason SkinPen by Crown Aesthetics is the world’s first FDA-cleared microneedling device. Learn more by reviewing our patient labeling document.
WHAT SHOULD YOU DO BEFORE A SKINPEN TREATMENT?
Before the SkinPen® Precision treatment, your doctor will ask you questions about your medical history, as well as your treatment goals. Your doctor will discuss whether you are an appropriate candidate for a SkinPen® Precision System treatment and review what to expect during and after a treatment, including common treatment responses and adverse events. You may be cautioned to avoid sun exposure and stop topical retinoid therapy 24 hours prior to procedure. You should allow at least 24 hours after autoimmune therapies before a treatment. You should wait six months following oral isotretinoin (such as Accutane) use before receiving treatment. Your doctor will also examine your treatment area, and may take photos. Different options for pain management will be discussed, and if pre-treatment numbing is desired, a topical anesthetic agent may be used. The treatment area will be cleaned and then prepared with isopropyl alcohol or other antiseptic before the treatment.
SCHEDULE AN APPOINTMENT TODAY
ADDRESS
711 E Lamar Blvd # 200
Arlington, TX 76011
moreinfo@acderm.com
PHONE
817-795-SKIN (7546)
HOURS
Mon – Thur | 7:00 AM – 4:00 PM
Fri | 07:00 AM – 12:00 PM
Closed on Weekends







